
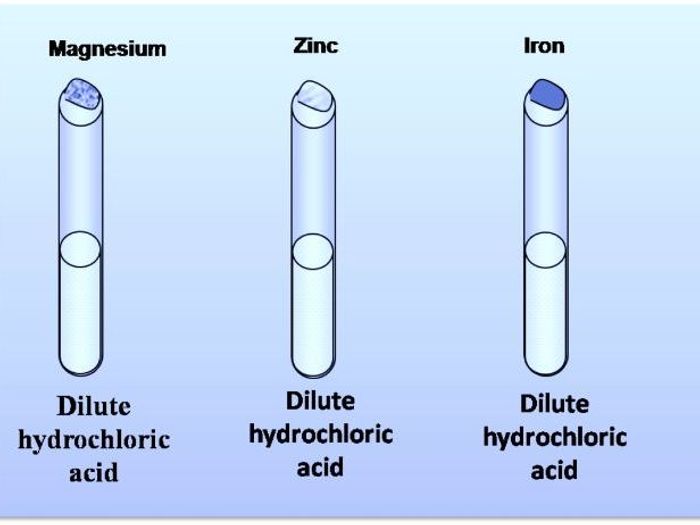

This can be remembered with the help of the following mnemonics. For anions, the hydroxide (OH) is the ion that. A single-displacement reaction, also known as single replacement reaction or exchange reaction, is a chemical reaction in which one element is replaced by another in a compound. Predicting and determining the products using the reactivity series. Cations are positively (+) charged ions while anions are negatively () charged. Reactivity Series: The cation of the metal lower down in the reactivity will be the first one to decompose.


 0 kommentar(er)
0 kommentar(er)
